
時間 : 2026年03月30日 10時00分
地點 : 能動學(xué)院301會議室
主講人 : Prof. Olaf Deutschmann
The transformation of the energy and chemical sectors from fossil fuels to renewable energy sources demands the development of long-term, large-scale chemical energy storage and transportation systems. This seminar will focus on alternative routes for storing renewable energy in the form of iron and ammonia.
Iron is a carbon-free, safe, scalable, grid-independent, and non-toxic material that shows great promise for storing and transporting renewable energy due to its high volumetric energy density [1]. In a cyclical process, renewable electricity can be stored by chemically reducing iron oxides using electrolytic hydrogen in sunny and windy regions or during periods of excess renewable electricity. The reduced metals can then be transported and used in regions with high energy demand but limited availability of renewable resources. Energy in the form of electricity, heat, and hydrogen can then be released on demand by thermochemical oxidation, either with air or steam. The cycle is completed when the metal oxides are regenerated where and when renewable electricity is available.
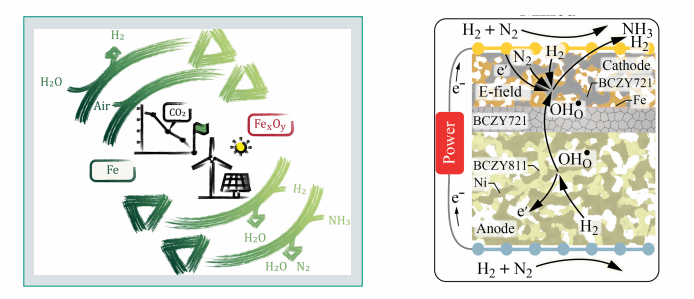
Sketches of storage-release energy cycles using iron [1] (left) and NH3 synthesis in PCCs [2] (right).
Ammonia can serve as a chemical energy carrier produced in a decentralized manner via electrocatalysis. Recent advances have been made particularly in proton-conducting ceramic cells (PCCs) using iron- and ruthenium-based electrodes [2]. A strong interaction between electro- and thermo-catalytic reactions occurs in such cells. Co-feeding H? at the cathode has proven advantageous for optimizing reaction conditions and increasing ammonia synthesis rates. Additionally, the decomposition of NH? to H? can be carried out in PCCs. Alternatively, NH? can be directly converted into electricity in solid-oxide fuel cells (SOFCs) [3].
[1] C. Kuhn et al., ChemSusChem (2024) DOI: 10.1002/cssc.202400351.
[2] P. Blanck et al., J. Electrochem. Soc. (2025) DOI: 10.1149/1945-7111/adfc9e.
[3] A. Ashar et al., Applied Energy (2025) DOI: 10.1016/j.apenergy.2025.125788
主講人簡介:
Professor Olaf Deutschmann holds the Chair of Chemical Technology at the Karlsruhe Instiute of Technology(KIT) in Germany. He also serves as director of the Institute for Chemical Technology and Polymer Chemistry, aswell as the Instiute of Catalysis Research and Technology at KIT. His research focuses on the development oclimate- and environment-friendly chemical technologies, along with novel digitalization tools. His current researchtopics include carbon capture, carbon-free chemical energy, emission control, fuel and electrolysis cells, and thecircular economy. Prof. Deutschmann studied physics and chemistry in Magdeburg, Berin and Heidelberg. He has published over 370 peer-reviewed journal papers with H-index 77, and deliverecmore than 100 invited lectures and seminars, including the Distinguished Lecture Series at UC Berkeley. He hasreceived the DECHEMA Award, the Hermann Oberth Medal, and is a Fellow of the Combustion Institute.

編輯:曹蔚
責(zé)編:韋麗